Free Case Evaluation
You will never be charged a fee unless a recovery is made for you.
We are no longer accepting new cases.
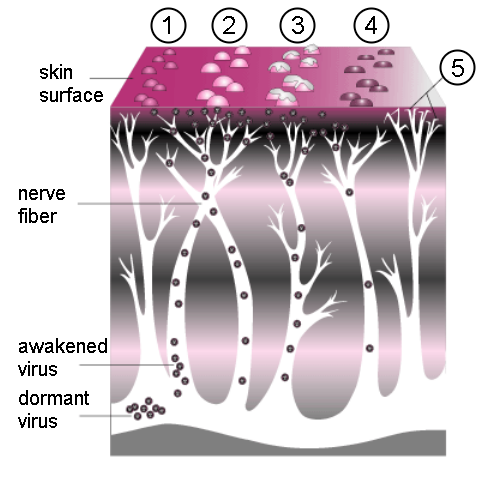
Progression of shingles. A cluster of small bumps (1) turns into blisters (2). The blisters fill with lymph, break open (3), crust over (4), and finally disappear. Postherpetic neuralgia can sometimes occur due to nerve damage (5).
Trustwell Law Group is now accepting cases on behalf of people who have contracted shingles or experienced other serious side effects after being vaccinated with Zostavax.
The Zostavax lawsuits claim that the vaccine manufacturer—Merck & Co.—learned during its clinical trials that the vaccine could cause patients to contract shingles but failed to disclose this danger. The Zostavax label did not list shingles as a possible side effect until late 2014, nearly seven years after the vaccine received FDA approval.
If you or a loved one were diagnosed with shingles within a year after receiving Zostavax, or experienced an adverse reaction such as neuralgia (severe nerve pain) or contracted an autoimmune disorder within two years after receiving Zostavax, call us at 800-796-1636 or submit your case details online. A member of our legal team will contact you shortly. You may be entitled to compensation.
Our attorneys have years of experience and a reputation for personalized, compassionate partnering with our clients. We also have access to the expertise, resources, and manpower to fully investigate your circumstances, file a lawsuit if appropriate, and help you seek the justice you deserve.

Zostavax—manufactured by Merck & Co Inc.—is a vaccine1 designed to reduce a person’s risk of contracting shingles. Shingles (herpes zoster) is an infection caused when dormant copies of the varicella zoster virus (which also causes chicken pox) reactivate in the patient’s body.
Zostavax is a “live-attenuated” vaccine2,3 meaning it contains a live but weakened form of the virus it is intended to guard against (the varicella-zoster virus). The vaccine is intended to stimulate the production of antibodies by the immune system to more effectively suppress the virus present in the patient’s body.
The FDA approved Zostavax in 2006 to prevent shingles in persons aged 60 or older without compromised immune systems. At the time, Zostavax was the only shingles vaccine approved in the United States. In 2011, the FDA expanded that approval to persons between the ages of 50 and 59. When approved, Zostavax was expected to be roughly 50% effective, reducing the incidence of shingles by half, based on a study of older adults given the vaccine.
However, some patients whose conditions worsened after receiving Zostavax have claimed the vaccine caused shingles and other serious side effects and that the Zostavax label hadn’t properly warned of those risks. In 2014, Merck added shingles as a potential side-effect listed in the Zostavax labeling.
Scientists at the Centers for Disease Control (“CDC”) analyzed data collected by the Vaccine Adverse Event Reporting System maintained jointly by the CDC and the FDA. They found more than 1,100 reports of “serious” adverse reactions in patients receiving Zostavax – more than 8 percent of which caused the patient’s death.
In 2017 the FDA approved a second shingles vaccine—Shingrix®, manufactured by GlaxoSmithKline Biologicals. Shingrix is a recombinant vaccine2,6 (made using the DNA from a portion (subunit) of the zoster virus) and contains no live virus.
In October 2017, the Advisory Committee on Immunization Practices of the CDC recommended patients receive Shingrix rather than Zostavax because the newer vaccine was expected to be safer and much more effective (91% efficacy).
Shingles is a painful, blistering rash appearing most often on one side of the body in older adults who have contracted or been exposed to chicken pox (varicella). In some shingles patients, chronic pain remains even after the rash has cleared (a condition called postherpetic neuralgia).
Chicken pox was once a nearly universal childhood illness in the United States, which has become less common since a chicken pox vaccine was approved in 1996. Studies show that more than 99% of Americans over the age of 40 have had chicken pox, even if they don’t remember it. After a person recovers from chicken pox, the zoster virus becomes dormant but remains within the nerve cells in the spinal cord and in the brain.
In adult life, this virus can become active again and cause shingles. Estimates from the Centers for Disease Control indicate that around one-third of the population will suffer from shingles at some point. Shingles primarily affects people 60 and older, but the virus can also become active again in younger individuals with compromised immune systems. People who contracted chickenpox before they were 18 months old are at elevated risk for shingles
The first symptom of shingles is a painful tingling or burning sensation at a specific location on the skin. A few days later, a rash may appear, and blistering can follow. An outbreak can last for up to a month. However, in roughly 20 percent of shingles cases, the patient experiences continuing nerve pain for months or years.
Shingles (herpes zoster) is one potential side effect from Zostavax. In addition to the painful skin rash, the shingles virus (varicella-zoster virus) can cause serious nerve damage, encephalitis, meningitis, and autoimmune disorders, and puts patients at risk for stroke, heart failure, and pneumonia.
Injuries linked to the shingles virus include:
Patients who have contracted shingles or experienced other serious side effects after being vaccinated with Zostavax have sued Merck & Co. seeking compensation for their injuries. These cases are at an early stage; there have been no outcomes announced (verdicts or settlements) in any lawsuit claiming injuries from Zostavax. The Zostavax lawsuits filed in multiple states have now been consolidated.
Mass tort litigations typically involves large groups of cases being combined in federal court for discovery and other pre-trial purposes in what is called a Multidistrict Litigation (“MDL”). That has happened in the Zostavax lawsuit. More than 50 cases filed against Merck & Co (and its parent company, Bayer AG) have been consolidated in the Eastern District of Pennsylvania to form the Zostavax MDL. The Zostavax MDL claims that:
Generally, MDLs do not settle until after several “bellwether” cases have been tried to a jury. The results of those trials allow the manufacturer and the plaintiffs to better understand the impact of the available evidence and the gauge the risks of continued litigation.
End Notes
Article Sources
You will never be charged a fee unless a recovery is made for you.